Installation of telerobotic surgery and initial experience with telerobotic radical prostatectomy
Anthony J. Costello, Hodo Haxhimolla, Helen Crowe and Justin S. Peters
OBJECTIVE
To assess the ability of untrained laparoscopic surgeons to learn and implement laparoscopic telerobotic radical prostatectomy (TRP) using the daVinci Surgical System (Intuitive Surgical, CA), and assess the education, safety and efficacy issues when instituting this system.
PATIENTS AND METHODS
Between December 2003 and October 2004, 122 consecutive TRPs were performed by two surgeons for clinically localised prostate cancer. The individual robotic surgeon was assisted at the bedside by another surgeon. The TRP was performed robotically by the surgeon at the remote console unit. Perioperative data and pathological results were recorded. The two surgeons spent 1 week in a skills laboratory using a porcine model of laparoscopic TRP, and then cadaveric robotic prostatectomy. The first six cases were mentored by an experienced telerobotic surgeon.
RESULTS
The TRP was conducted by two surgeons with no previous laparoscopic experience. There were no conversions to open surgery. Assessing the complications, postoperative continence, operating time and transfusion rates showed equivalent efficacy and safety to open and pure laparoscopic methods.
CONCLUSION
TRP represents a novel computer-based surgical approach to prostate cancer, which offers the benefits of minimally invasive surgery without the extensive experience associated with the laparoscopic method. It remains to be seen whether the robotic approach can deliver better outcomes in continence and potency over time.
INTRODUCTION
elerobotic surgery allows a closed laparoscopic abdominal approach, placing a computer between the patient and surgeon. The surgeon's hand movements are digitised to improve dexterity. The system has the added benefit of three- dimensional visualisation compared to the conventional laparoscopic approach. Pure laparoscopy is counter-intuitive compared with telerobotic radical prostatectomy (TRP), which is intuitive for the surgeon. Robotic surgery is a beguiling surgical innovation and some of the enthusiasm during installation of robotic systems relates to maintenance or increasing surgical market share. The novelty of the technology means that it is at present unproven, with high capital cost. The DaVinci Surgical Robotic System (Intuitive Surgical, CA, USA) is a master slave telemanipulation system (Fig. 1). The master-slave system consists of a remote console where the operating surgeon (master) directs the robotic surgical arms (slave) via a telerobotic videoscopic link. The DaVinci system represents an important technological breakthrough. It has transformed conventional laparoscopic surgery from a two-dimensional counter intuitive procedure to a fully intuitive natural surgical procedure using excellent visualisation. Previous laparoscopic surgery has some advantages over open approaches for RP. These relate to reduced pain, early discharge and early return to normal activity. Laparoscopic TRIP has the potential to improve patient outcomes compared with open RP. Tangible benefits relate to improve visualisation via pneumoperitoneum, which also provides tamponade reducing the intraoperative bleeding. The absence of an abdominal incision means less postoperative pain, improved cosmesis and early discharge. At present there is no evidence to suggest that there is any improvement in the rates of return to urinary continence, and it is too early to determine whether improved surgical dexterity and visualisation will actually improve postoperative potency. This is a key area where technology may improve the results [1]. The need for transfusion is much reduced in our TRP series compared to our open series. Three patients in the first 100 required a blood transfusion. Historically in our open RP series, 60% of patients usually had an autologous transfusion. There are reports worldwide with much lower open transfusion rates than ours [1]. However, in our hands one of the remarkable advances of laparoscopic prostatectomy relates to haemostasis and reduced blood loss. To establish a functioning telerobotic surgical service, ideally it should be multidisciplinary. Significant training requirements were necessary before establishing the service for operating room nurses and technicians, and engineering staff responsible for maintaining the equipment. The operating room had to be reconfigured. Cardiovascular surgeons have also embraced the technology, mainly for the repair of mitral valves and atrial septal defects. Surgeons who are skilled open surgeons can transfer their skills very easily to a telerobotic laparoscopic approach. There appears to be no requirement for previous general laparoscopic skills [2]. Certainly a single-team approach with two surgeons and consistent table-side assistance, and trained operating room nursing staff, has made the institution of this programme much easier [3].
FIG 1. The components of the DaVinci System
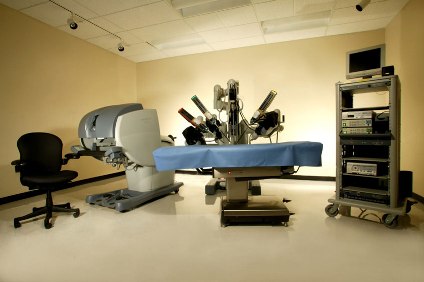
ELEMENTS OF THE DA VINCI SURGICAL SYSTEM
The surgical console provides the computer interface between surgeon and surgical robotic arms. The surgeon controls the robotic arms through the use of master handles, which are located in virtual three-dimensional space below the visual display. The surgeon's hand movements are digitised and transmitted to the robotic arms, which perform in identical movements in the operative field. Foot controls are used to activate electrocautery, for repositioning the master handles and for focusing. The surgeon views the surgical field through the binocular display in the hood of the console. The robotic arms are deactivated when the surgeon's eyes are removed from the display. The surgeon's console and the robotic-arm cart are connected via a data cable. In the USA, Food and Drug Administration approval for this technology mandates that the operating surgeon is in the same room as the patient. However tele-surgery in which the patient and surgeon are remote is possible, and has been reported [4].
MASTER HANDLE
In addition to providing direction to the robotic arms, the master handles are also used to control other aspects of the video display system and robotic arms, such as endoscope selection and motion-scaling ratio. The master handles filter tremor in the surgeon's hands and arms (Fig. 2). The majority of tactile feedback is provided indirectly by the video monitor, that is visually, and the tensile feedback through the robotic arms. The robotic-arm cart is placed beside the patient on the operating table. It holds three, or more recently four, robotic arms on a central tower. One arm holds the videoscope and the others are used to attach instrument adapters which are connected to robotic instrumentation through reusable trocars. Stereoscopic vision is supplied by a 30° or 0° specialised three-dimensional endoscope, which provides the surgeon at the console with binocular vision in the operative field.
FIG 2. The DaVinci Master handle

The robotic surgical instruments have both an elbow joint and wrist, enabling seven degrees of freedom and two degrees of axial rotation, mimicking the natural motions of open surgery. This is in contrast to conventional laparoscopic surgery, where the surgeon's hand movements are counter-intuitive and in two dimensions. There is a range of different instruments available which can be used up to 10 times, after which the robotic system deactivates them and prevents further use.
PATIENTS AND METHODS
In all, 122 men (mean age 61.2 years, range 48-72) underwent TRP by two surgeons between December 2003 and December 2004. Information on continence after TRP was collected by questionnaires sent to all patients, with a return stamped, self-addressed envelope included.
SURGICAL TECHNIQUE
The technique of TRP was adapted from that described by surgeons using a purely laparoscopic approach [5,6]. A protocol of surgical steps was used in all the present patients, and all were transperitoneal. The technique we adapted was originally described as the Montsouris technique, modified at the University of California Irvine. The same surgical steps are used in all cases: (i) Establishing pneumoperitoneum via Hassan cannula (used in preference to Veress needle after the first 30 cases); (ii) placing the trocars; (iii) docking the robot; (iv) taking down the urachus and defining the space of Retzius for dividing the superficial dorsal veins of the penis; (v) incision of the lateral pelvic fascia; (vi) dividing the puboprostatic ligaments; (vii) staple ligation of the dorsal venous complex; (viii) dividing the junction of the bladder neck and prostate; (ix) dividing the fascial layer above the seminal vesicles, with dissection of the vasa deferentia and control of the blood vessels supplying these structures; (x) dividing the anterior layer of Denonvilliers' fascia, exposing the anterior wall of the rectum; (xi) dividing the prostatic pedicles; (xii) dissecting bilaterally the neurovascular bundles; (xiii) dividing the prostate and urethra at the apex; (xiv) removing the prostate in an endoscopic bag. This technique has been used in all but one patient at our centre; in the one case, the dissection had to be retrograde, from apex of the prostate to bladder neck, because of difficulty with rectal dissection. The modifications to the technique in the present series relate to the use of a suprapubic needle to the lasso in the Foley catheter after dividing the bladder neck. The needle is passed through the eye of the catheter with a one Nylon suture, which is then brought out suprapubically to add traction to the prostate anteriorly. Two other surgical ports are placed, one in the left iliac fossa and one below the left costal margin. These ports are used by the bedside surgeon for instrumentation being suction, irrigation and surgical retraction.
RESULTS
No patient required conversion to open surgery. The mean (range) preoperative PSA level was 8.4 (1.2-25) ng/mL, the prostatic volume 44.7 (20-106) mL and the body mass index 27.2 (20.2_38.1) kg/m2. The clinical and pathological T stage is shown in Table 1. The mean (range) stay after TRP was 2 (1-9) days, and the indwelling catheter time 8.4 (5-33) days (median 7). The margin status is shown in Table 2; the overall positive margin rate (tumour at the inked margin) was 16.3%, including six patients who had positive seminal vesicle involvement. Data were available on urinary continence in 93 patients at 3 months (Table 3); four patients were incontinent before TRP and wore pads, thus they were excluded from the analysis of continence after TRP. Only one patient declined to complete the continence questionnaire. At 3 months, 65 patients (73%) reported they were pad-free or wearing one 'security' pad; by 6 months 82% of patients were continent. Preliminary data were available for erectile function but were too premature for a meaningful assessment of long-term erectile dysfunction after TRP. It may take up to 2 years for the return of erectile function after nerve-sparing robotic RP. Four patients (3%) received blood transfusions; other complications are listed in Table 4.
Table 1. Clinical and pathological staging of the 122 men
Table 3. The frequency of incontinence during the follow-up, and pad use/24h
DISCUSSION
TRP was popularised and championed at the Vattikuti Urology Institute by Menon and Tewari [7] and Tewari et a/. [8]. The present report shows the replacement of an open operation with TRIP. Laparoscopic RP has an equivalent oncological outcome to reported open series [9]. Weider and Soloway [10] reported overall positive margin rates of 28%; those for laparoscopic RP are reportedly 19-23% [11,12]. The morbidity (safety) of this new procedure would appear to be at least equivalent to the experience in major centres with open surgery [11]. The rate of return to continence at 6 months was 82%, with patients using no or one pad per day, which would appear to be acceptable. Further follow-up beyond a year for both continence and erectile function is necessary and underway. The true benefits of this procedure over open RP clearly relate to reduced blood loss, absence of abdominal incision, early discharge and early return to normal activity. Nerve sparing was attempted in almost all patients (step xii). The vision system allows excellent visualisation of the neurovascular bundles. Robotic technology has long been present in industry but only recently has it been an option for surgeons [13]. A cholecystectomy was conducted between New York and Paris by telerobotic means [4]. The Zeus System has been trialled for several years, as a voice-activated surgical robot. We think that the introduction of telerobotic laparoscopic surgery is a watershed in surgical development, and constitutes a major technological advance in minimally invasive surgery. Laparoscopy has confirmed benefits for reduction in length of stay, absence of incision and early return to normal activity, reduced infection, improved cosmesis and possibly less interference with the immune response. A major advance provided by 'intuitive' robotics vs. laparoscopy is that the robotic approach allows the surgeon's natural hand-eye coordination and a natural enhanced dexterity. This contrasts with the two-dimensional counter-intuitive reverse-hand movement of pure laparoscopy. Many surgeons have found the transition to laparoscopy difficult. The new minimally invasive surgeon with little or no laparoscopic experience can quickly adapt to the laparoscopic approach to RP using robotics. The ability to view the surgical field in three dimensions using natural hand and arm movements, and the use of filters for hand and arm tremor, is significant. The addition of motion scaling, such that large movements are reduced to fine movements, is an advantage. The robotic system removes surgical tremor, which is compensated on the end motion by computer filters. A further advantage to the surgeon is improved visualisation via the three-dimensional camera system, which has x 10 magnification in a more appropriate comfortable ergonomic environment. The three-dimensional magnified view is a dramatic improvement over conventional two-dimensional laparoscopic visualisation. Pelvic open surgery for retropubic RP requires the surgeon and assistant to adopt sometimes anatomically difficult positions, stressing the cervical and lumbar spines. Benefits are conferred to the operating surgeon as a result of the ergonomic set-up of the surgeon console. Retropubic RP is a difficult open surgical operation and lends itself to the telerobotic laparoscopic approach, with improved dexterity and visualisation in an anatomically confined area of subpubic access; it is macrosurgery performed in a microsurgical fashion. These advantages perhaps outweigh the clear problems of the shift to robotics. The high capital costs, lack of compatible instrumentation, large physical size of the robot and eventual obsolescence are obviously concerns to be addressed over time. If robotics are expected to be embraced widely, exciting additional advances could ensue. The overlay of MRI and CT images for surgical guidance, and the addition of haptic feedback, are potentially feasible. The application of telerobotics across all surgical disciplines is likely [14] and application of this system is limited only by the surgeon's imagination. In urology TRP now seems to have an enduring position. Further applications in urology will relate to partial nephrectomy, cystoprostatectomy [15], pyeloplasty and ureterolysis.
CONFLICT OF INTEREST
None declared.
REFERENCES
1. Herrell SD, Smith JA. Laparoscopic and robotic radical prostatectomy: what are the real advantages? BJU Int 2005; 95: 3-4
2. Ahlering TE, Skarecky D, Lee D, Clayman RV. Successful transfer of open surgical skills to a laparoscopic environment using a robotic interface: Initial experience with laparoscopic radical prostatectomy. J Uro12003; 170: 1738-41
3. Steers WD, LeBeau S, Cardell J, Fulmer B. Establishing a robotics program. Urol Clin 2004; 31: 773-80
4. Challacombe BJ, Kavoussi LR, Dasgupta P. Trans-oceanic telerobotic surgery. BJU Int 2003; 92: 678-80
5. Guillonneau B, Vallancien G. Laparoscopic radical prostatectomy: The Montsouris technique.
6. Guillonneau B, EI-Fettouh H, Baumert H et al. Laparoscopic radical prostatectomy: Oncological evaluation after 1,000 cases at Montsouris Institute. J Uro12003; 169: 1261-6
7. Menon M, Tewari A. Vattikuti Institute Prostatectomy Team. Robotic radical prostatectomy and the Vattikuti Urology Institute technique: an interim analysis of results and technical points. Urology 2003; 61 (Suppl. 1): 15-20
8. Tewari A, Srivasatava A, Menon M. A prospective comparison of radical retropubic and robot-assisted prostatectomy: experience in one institution. BJU Int 2003; 92: 205-10
9. Thomas K, Slabaugh JR, Marshall F. A comparison of minimally invasive open and laparoscopic radical retropubic prostatectomy. J Uro12004; 172: 2546-8
10. Wieder JA, Soloway MS. Incidence, etiology, location, prevention and treatment of positive surigical margins after radical prostatectomy for prostate cancer. J Urol 1998; 160: 299-315
11. Stolzenburg JU, Do M, Rabenalt R et al. Endoscopic extraperitoneal radical prostatectomy: initial experience after 70 procedures. J Uro12003; 169: 2066-71
12. Rassweiler J, Zeeman O, Schulze M, Tebr D, Hatzinger M, Frede T. Laparoscopic versus open radical prostatectomy: a comparative study at a single institution.
13. Lanfranco AR, Castellanos AE, Jaydev MD, Desai P, Meyers MD. Robotic surgery: a current perspective.
14. Darzi A, Mackay S. Recent advances in minimal access surgery.
15. Balaji KC, Yohannes P, McBridge CL, Oleynikov D, Hemstreet GP. Feasibility of robot-assisted totally intracorporeal laparoscopic ileal conduit urinary diversion: Initial results of a single institutional pilot study. Urology 2004; 63: 51-5
BJU International
Volume 96 Issue 1 Page 34 - July 2005
